
BIGBEAR
PHARMACEUTICAL
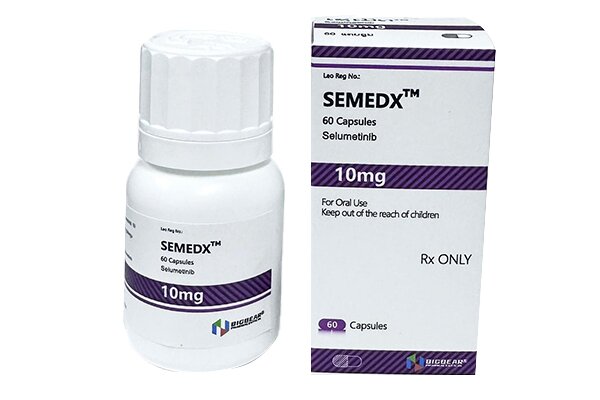
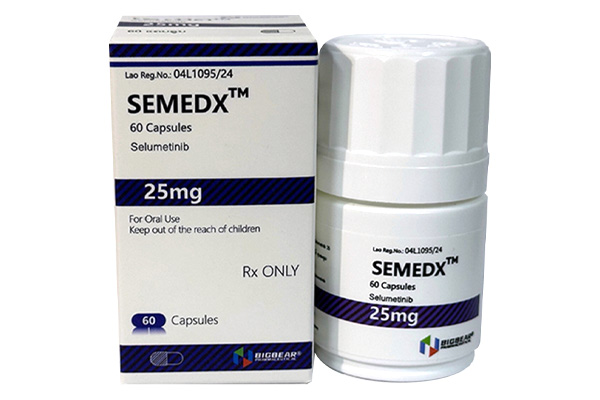
Selumetinib inhibits the activity of MEK1/2 proteins, blocks the RAS/RAF/MEK/ERK signaling pathway, and thereby suppresses the proliferation and survival of tumor cells.

 Authentic
Authentic Guarantee
Guarantee Fast Delivery
Fast Delivery Privacy
Privacy Neurofibromatosis type 1 (NF1) is an incurable genetic disorder that typically manifests in early childhood. Its severity varies greatly and can reduc···【More】
Update: 11 Mar,2026Source: BigbearViews: 96
The U.S. Food and Drug Administration (FDA) approved on September 10 the oral selective MEK inhibitor Koselugo (selumetinib) in oral granule and capsu···【More】
Update: 11 Mar,2026Source: BigbearViews: 91
On November 19, 2025, AstraZeneca announced that the U.S. Food and Drug Administration (FDA) has approved its mitogen-activated protein kinase kinase ···【More】
Update: 11 Mar,2026Source: BigbearViews: 91
FDA Approval AnnouncementOn November 19, 2025, AstraZeneca announced that the U.S. Food and Drug Administration (FDA) has approved Koselugo (selumetin···【More】
Update: 11 Mar,2026Source: BigbearViews: 92
FDA Approval AnnouncementOn September 10, the U.S. Food and Drug Administration (FDA) approved Koselugo (selumetinib) oral granules and capsules, an o···【More】
Update: 11 Mar,2026Source: BigbearViews: 92
Copyright2024@ BIGBEAR All right reserved Bigbear | Bigbear Pharmaceutical | Bigbear Laos



